Which of the Following Has the Shortest Bond Length
Which of the following has the shortest bond length. Answer 1 of 7.
Which Of The Following Has The Shortest Carboncarbon Class 12 Chemistry Cbse
Which of the following has the shortest bond length.

. Which diatomic molecule has the shortest bond length. H3C-H H2N-H HO-H F-H. Get Answer to any question just click a photo and upload the photo and get the answer completely free UPLOAD.
NCERT chemistry Part 1 class 11. Show transcribed image text Expert Answer. We can draw the structure of CO as In.
D resonance E van der Waals forces London dispersion forces 13. B-F CB-C ODB-O. That means more the bond order shorter the bond length.
A hydrogen bonding B hybridization C ionic bonding. Therefore the option C is correct. Who are the experts.
Which one of the following has the shortest bond length. This problem has been solved. See the answer See the answer See the answer done loading.
N O2 N O 2. Which diatomic molecule has the shortest bond length. CO NO CC CN is the bond length order Source.
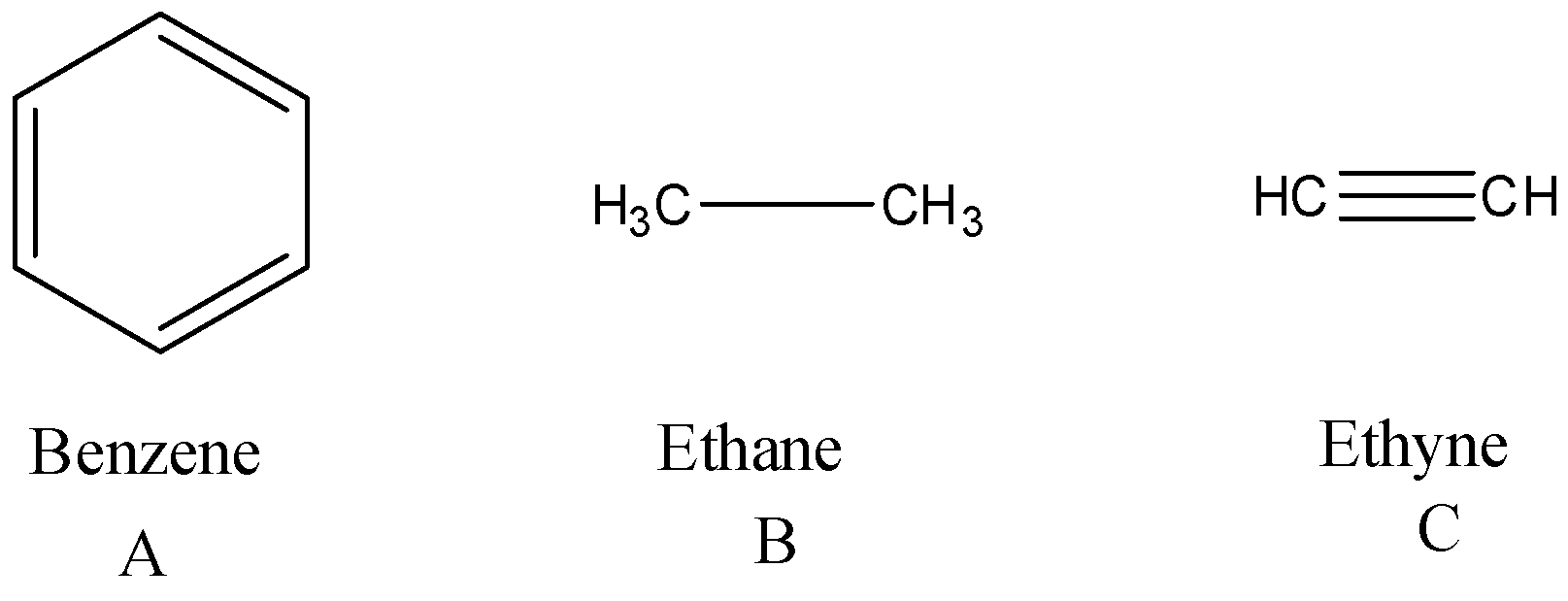
This problem has been solved. Here ethyne has a triple bond benzene has a double bond and ethane has a single bond. NO NO NO 2NO -.
Thus shorter will be the Bond length. Which one of the following has the shortest bond length. Which of the following would have the shortest bond length according to MO theory.
Is used to explain why the boiling point of HF is greater than the boiling point of HBr. The correct option is B. Benzene has more bond length than ethyne as it has bond order 2 and the highest bond length found is ethane as it has bond order 1.
O2 O22 O2- O22-. Therefore its the shortest bond length of C O bond. 1 Answer 1 vote.
Who are the experts. C 2 H 2 ethyne or acetylene due to presence of carbon-carbon triple bond has shortest carbon-carbon bond length. That is why.
The bonding MO has lower energy and hence greater stability whereas anti-bonding MO has more energy and hence lesser stability. This browser does not support the video element. Therefore option A is correct.
We review their content and. A O 2 B O-2 C O 2 D O-2 2. Correct answer is D O-2 2.
Show transcribed image text Expert Answer. Which of the following species has the shortest bond length. Thus N O N O has the highest bond order and therefore the.
Is used to explain the fact that the four bonds in. NCERT DC Pandey Sunil Batra HC Verma Pradeep. See the answer See the answer See the answer done loading.
C O bond gets triple bond character in one in all the resonating structures. Yes bond order will give an order which is different from what is observed but this is chemistry and experimental values do be a b sometimes. Which of the following species has the shortest bond length.
C 2 H 2. So the compound whose bond length is smallest is ethyne as it has bond order 3. Please answer BOTH questions and show workprocess.
Which of the following has the shortest bond length. Which of the following has the shortest bond length. Rank the bond lengths in the following molecule from shortest to longest.
Which of the following bonds has the shortest bond length. Answered Dec 27 2019 by komal01 247k points selected Dec 27 2019 by Manoj03. As O_22 has the highest bond order bond length will be small.
Share It On Facebook Twitter Email. 5 H 3 2 1 H Shortest 1 2 3. Which of the following would have the shortest bond length according to MO theory.
Which one of the following has the shortest bond length. Which of the following has the shortest bond length. One electron is removed from π 2px π 2 p x of NO Bond order 1 2 8 4 2 1 2 8 - 4 2.
Experts are tested by Chegg as specialists in their subject area. Now we can draw the structure of C H 3 O H as However in A exceedingly C O has a single bond character. O 2 BO.
Which one of the following has the shortest bond length.

Which Of The Following Has The Shortest Bond Length Youtube

Solved Which Of The Following Bonds Has The Shortest Bond Chegg Com
No comments for "Which of the Following Has the Shortest Bond Length"
Post a Comment